Hypertension in patients with coronavirus disease 2019 (COVID-19): a pooled analysis
Key words: coronavirus, COVID-19, hypertension



 CC BY-NC-SA 4.0
CC BY-NC-SA 4.0
Hypertension in patients with coronavirus disease 2019 (COVID-19): a pooled analysis
Introduction: As the outbreak of coronavirus disease 2019 (COVID‑19) was recognized, the clinical predictors of severe or fatal course of the disease should be identified to enable risk stratification and to allocate limited resources optimally. Hypertension has been widely reported to be associated with increased disease severity; however, some studies reported different findings.
Objectives: The study aimed to evaluate the association between hypertension and severe and fatal COVID‑19.
Methods: The Scopus, Medline, and Web of Science databases were searched to identify studies reporting the rate of hypertensive patients in the population diagnosed with severe or nonsevere COVID‑19 or in COVID‑19 survivors and nonsurvivors. The obtained data were pooled into a meta‑analysis to calculate odds ratios (ORs) with 95% CIs.
Results: Hypertension was associated with a nearly 2.5‑fold increased risk of severe COVID‑19 (OR, 2.49; 95% CI, 1.98–3.12; I2 = 24%), as well as with a similarly significant higher mortality risk (OR, 2.42; 95% CI, 1.51–3.90; I2 = 0%). In a meta‑regression analysis, a correlation was observed between an increase in the mean age of patients with severe COVID‑19 and an increased log OR of hypertension and COVID‑19 severity (P = 0.03).
Conclusions: This pooled analysis of the current literature would suggest that hypertension may be associated with an up to 2.5‑fold higher risk of severe or fatal COVID‑19, especially in older individuals.
What's new?
Hypertension has been widely reported to increase in severity in patients with coronavirus disease 2019 (COVID‑19) and is related to higher mortality in this population. However, early studies on COVID‑19 showed mixed findings with respect to hypertension. We aimed to assess the relationship between COVID‑19 and hypertension in a pooled analysis of early reports on COVID‑19. We found that hypertension is associated with an approximately 2.5‑fold higher risk of both increased severity and mortality. In a meta‑regression analysis, we observed that this effect was mainly seen in patients over the age of 60 years. As more data are needed in this field, we encourage medical professionals to adhere to more stringent public health precautions when treating patients with hypertension. Hypertension should be considered a clinical predictor of disease severity in older patients with COVID‑19.
Introduction
The outbreak of coronavirus disease 2019 (COVID‑19) has spread across the world, affecting over 500000 people and causing more than 25000 deaths to date.1 This infectious disease is caused by the severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2),2 which enters cells through the angiotensin‑converting enzyme 2 (ACE2) receptor and, therefore, human‑to‑human transmission occurs.3 The function of this enzyme is to catalyze the conversion of angiotensin II to angiotensin 1–7, a peptide which opposes the proinflammatory, pro‑oxidative, vasoconstrictive, and fibrotic properties of angiotensin II.4
Due to the interplay between SARS‑CoV‑2 and ACE2, it was suggested that hypertension may be involved in the pathogenesis of COVID‑19 either by playing a direct role as a pre‑existing clinical predictor of disease severity or by contributing to deterioration late in the course of the disease characterized by acute respiratory distress syndrome (ARDS), systemic inflammatory response syndrome (SIRS), and/or multiple organ failure (MOF).5 Considering the current virtually unstoppable trajectory of SARS‑CoV‑2,6 together with the high prevalence of hypertension (an estimated 26% of the world population),7 we may predict that the combination of these 2 conditions will soon pose overwhelming clinical, societal, and economic burdens on humans.8 However, whereas the media widely reported that hypertension increases the risk of severe COVID‑19, some early reports found no association between hypertension and disease severity.9,10 Therefore, this article aims to examine the strength of a possible clinical interplay between hypertension and COVID‑19 in order to learn whether hypertensive patients infected with SARS‑CoV‑2 may be at particularly increased risk of the worst clinical outcomes.
Methods
Search strategy and study selection
A systematic search was carried out in the Scopus, Medline (through the PubMed interface), and Web of Science databases, using the free key words “hypertension” OR “coronavirus disease 2019” OR “COVID‑19” OR “SARS‑CoV‑2” in all fields, including data published to March 26, 2020. We imposed no language restrictions. Additionally, the references of all included studies were hand‑searched to identify other possibly eligible studies. The articles that fulfilled our search criteria were then carefully assessed by title, abstract, and full text, if available. Our study was performed in compliance with the Declaration of Helsinki and local legislation. No ethics committee approval was necessary. The Preferred Reporting Items for Systematic Reviews and Meta‑Analyses (PRISMA) guidelines were followed (Supplementary material).
Each study was evaluated by 2 reviewers for inclusion. Disagreement between them was solved by consensus of all authors. All studies reporting on the prevalence of hypertension in adult (ie, aged 18 years or older) patients with laboratory‑confirmed, severe or nonsevere COVID‑19 or on COVID‑19 survivors and nonsurvivors were eligible for a pooled analysis. Furthermore, the criteria of the clinically validated definition of “severe disease” (ie, a condition in which patients experience severe respiratory distress, need mechanical ventilation or vital life support, or must be admitted to an intensive care unit) had to be met. Due to the limited literature on COVID‑19, no exclusion criteria were applied.
Data collection
The 2 reviewers independently collected data from the included studies. Prior to data collection, the articles written in Chinese were translated by a medical professional fluent in both Chinese and English. Data were collected and entered into a spreadsheet. Variables included authors, sample size, patient age, adopted definition of disease severity, patient outcomes, and prevalence of hypertension. No methodological risk of bias or publication bias were assessed, as the expected data set was limited.
Statistical analysis
A pooled analysis was performed to estimate the odds ratio (OR) and 95% CI of hypertension in patients with severe or nonsevere COVID‑19, or in COVID‑19 survivors versus nonsurvivors. The analysis was carried out with the MetaXL software, version 5.3 (EpiGear International Pty Ltd., Sunrise Beach, Australia), using an inverse‑variance model. Heterogeneity was evaluated using the χ2 test and the I2 statistic. For the χ2 test, significant heterogeneity among studies was indicated with a P value less than 0.1 obtained in the Cochran’s Q test. The results of the I2 statistic were interpreted as 25%, 50%, and 75%, representing low, moderate, and high heterogeneity, respectively. A leave‑one‑out sensitivity analysis was employed to probe the source of heterogeneity. Moreover, a random effects meta‑regression analysis using log OR was applied to evaluate the impact of mean age on the association between hypertension and the severity of COVID‑19 in severely ill patients.
Results
Search results and study characteristics
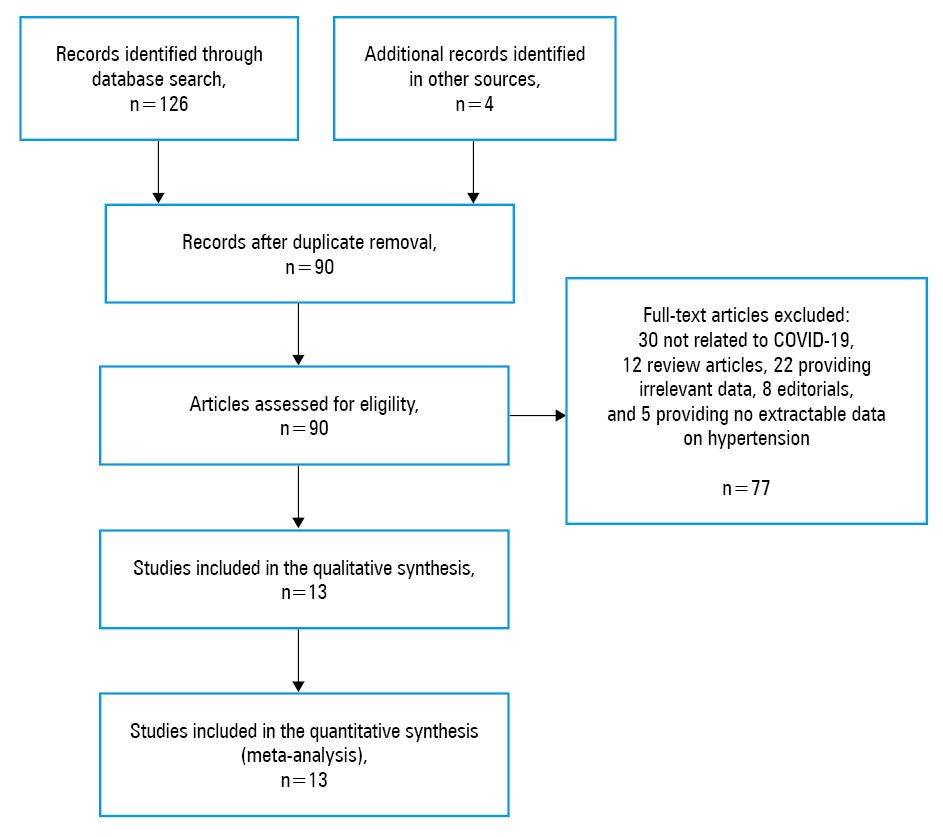
The flow chart of the analysis is presented in Figure 1. Overall, based on our electronic and reference search and following duplicate removal, 86 articles were initially identified as eligible for the study, 77 of which were excluded after screening by title, abstract, and full text: 30 articles were not related to COVID‑19, 12 were review articles, 22 did not provide relevant data, 8 were editorials, and 5 did not provide extractable data on hypertension. Four studies from the reference search were identified as eligible. Thus, 13 studies,9-21 with a total of 2893 patients with COVID‑19, were finally included in our analysis. Eleven studies compared the prevalence of hypertension in 2552 patients with confirmed severe versus nonsevere COVID‑19, 748 of whom (29.3%) were classified as having developed severe COVID‑19.9-19 Only 2 studies compared the prevalence of hypertension in 219 COVID‑19 survivors (64.2%) and 122 nonsurvivors (35.8%).20,21 The essential characteristics of the included studies are shown in Table 1.
Study | Location | Sample size | Patient outcomes | Patients with severe COVID‑19 | Patients with nonsevere COVID‑19 | ||||
n (%) | Agea, y | HTN, n (%) | n (%) | Agea, y | HTN, n (%) | ||||
a Data presented as median (interquartile range) or mean (SD)
Abbreviations: HTN, hypertension; ICU, intensive care unit; MV, mechanical ventilation; SpO2, oxygen saturation by pulse oximetry; others, see Figure 1 | |||||||||
Chen et al17 | China | 150 | Respiratory distress/insufficiency | 24 (16) | 68.5 (13.6) | 14 (58.3) | 126 (84) | 51.1 (15.6) | 35 (27.8) |
Guan et al11 | China | 1099 | Admission to ICU, MV, death | 173 (15.7) | 52 (40–65) | 41 (23.7) | 926 (84.3) | 45 (34–57) | 124 (13.4) |
Huang et al9 | China | 41 | ICU care | 13 (31.7) | 49 (41–61) | 2 (15.4) | 28 (68.3) | 49 (41–58) | 4 (14.3) |
Liu et al14 | China | 78 | Admission to ICU, MV, death | 11 (14.1) | 66 (51–70) | 2 (18.2) | 67 (85.9) | 37 (32–41) | 6 (9) |
Ruan et al20 | China | 150 | Death | 68 (45.3) | 67 (15–81) | 29 (42.6) | 82 (54.6) | 50 (44–81) | 23 (28) |
Qin et al12 | China | 452 | Respiratory distress/insufficiency | 286 (63.3) | 61 (51–69) | 105 (36.7) | 166 (36.7) | 53 (41.25–62) | 30 (18.1) |
Wan et al10 | China | 135 | Respiratory distress/insufficiency | 40 (29.6) | 56 (52–73) | 4 (10) | 95 (70.4) | 44 (33–49) | 9 (9.5) |
Wang et al16 | China | 138 | Clinical variables, MV, death | 36 (26.1) | 66 (57–78) | 21 (58.3) | 102 (73.9) | 51 (37–62) | 22 (21.6) |
Wang et al15 | China | 69 | SpO2 <90% | 14 (20.3) | 70.5 (62–77) | 5 (35.7) | 55 (79.7) | 37 (32–51) | 4 (7.3) |
Wu et al13 | China | 201 | ARDS | 84 (41.8) | 58.5 (50–69) | 23 (27.4) | 117 (58.2) | 48 (40–54) | 16 (13.7) |
Xiang et al18 | China | 49 | Respiratory distress/insufficiency | 9 (18.4) | 53 (14) | 4 (44.4) | 40 (81.6) | 40.6 (14.3) | 2 (5) |
Zhang et al19 | China | 140 | Respiratory distress/insufficiency | 58 (41.4) | 64 (25–87) | 22 (37.9) | 82 (58.6) | 52 (26–78) | 20 (24.4) |
Zhou et al21 | China | 140 | Death | 54 (28.3) | 69 (63–76) | 26 (48.1) | 137 (71.7) | 52 (45–58) | 32 (23.4) |
Meta‑analysis
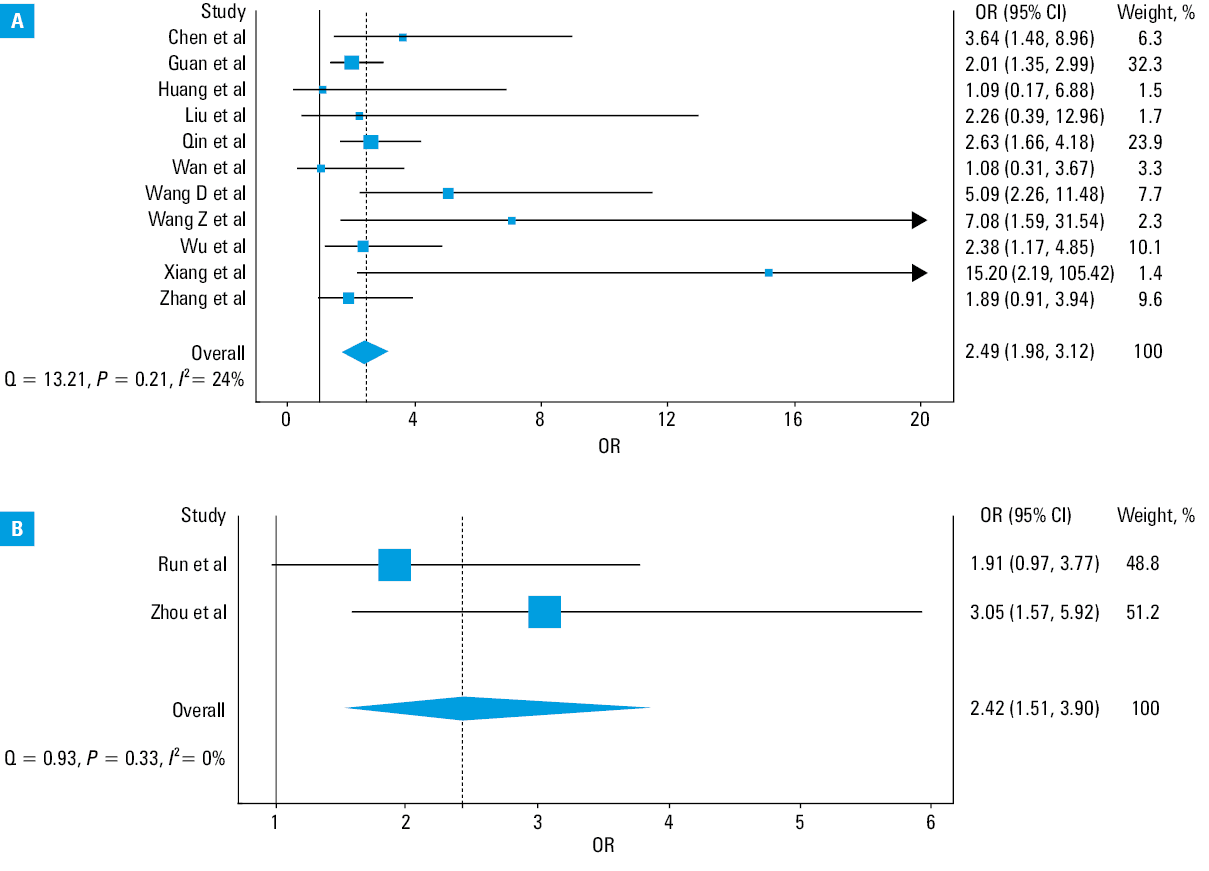
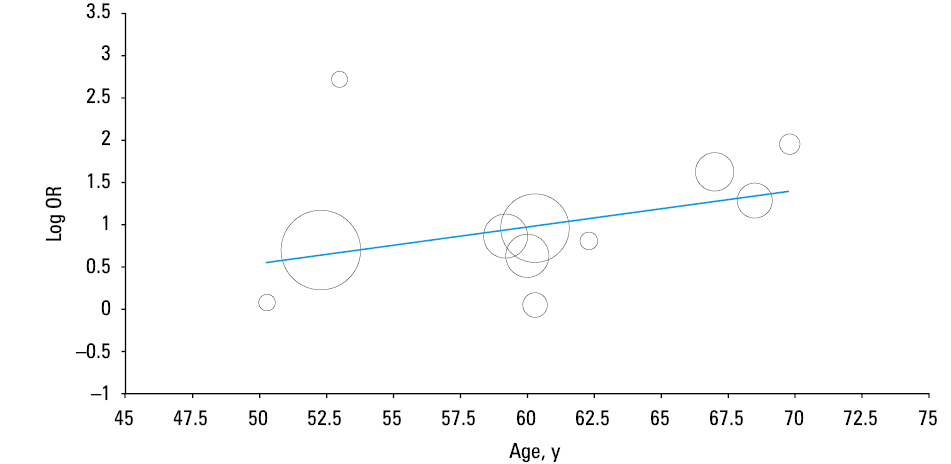
The results of our pooled analysis are presented in Figure 2. Hypertension was found to be associated with a nearly 2.5‑fold increased risk of severe COVID‑19 (OR, 2.49; 95% CI, 1.98–3.12; I2 = 24%; Cochran’s Q, P = 0.21), as well as with a similarly significant higher mortality risk (OR, 2.42; 95% CI, 1.51–3.9; I2 = 0%; Cochran’s Q, P = 0.33). A minimal heterogeneity was identified in the analyses. No significant differences were observed in the leave‑one‑out sensitivity analysis for disease severity (data not shown). In a meta‑regression analysis, a significant correlation was found between the mean age of patients with severe COVID‑19 and the log OR of hypertension and severity of COVID‑19 (correlation coefficient, 0.04; P = 0.03) (Figure 3).
Discussion
As the COVID‑19 pandemic continues, there is an increasing risk of overwhelming healthcare facilities and jeopardizing patient care even in the most developed countries. In this situation, reliable demographic, clinical, and laboratory indicators should be identified to distinguish patients with COVID‑19 who are at increased risk and, thus, need more aggressive management, including hospitalization or intensive care, from those who could be safely managed on an outpatient basis. Some laboratory parameters which may predict worse disease progression have already been identified, including leukocytosis, lymphopenia, thrombocytopenia, along with increased values of D‑dimers, procalcitonin, cardiac biomarkers, proinflammatory cytokines, and ferritin.22 Of note, some clinical predictors of worse prognosis in COVID‑19 were also reported in early studies, such as older age, male sex, as well as preexisting cardiovascular diseases, diabetes, respiratory disorders, cancer, and dementia.21,23 These findings are supported by observations regarding other respiratory and systemic diseases, as having at least one such comorbidity is now universally recognized to be an unfavorable prognostic factor in patients with pneumonia of various etiologies,24 ARDS,25 and SIRS.26 However, the strength of the association between these comorbidities and an increased risk of severe COVID‑19 has not been established yet.
In this study, we observed that hypertension carries a nearly 2.5‑fold higher risk of developing severe COVID‑19 or dying of SARS‑CoV‑2 infection (Figure 1). Although this association seems weaker than that reported earlier for other comorbidities, such as chronic obstructive pulmonary disease (over 5‑fold higher risk)27 or chronic kidney disease (over 3‑fold higher risk),28 it still carries important clinical implications.
As previously discussed, SARS‑CoV‑2 enters human cells by binding to the ACE2 receptor. Interestingly, previous studies showed that some antihypertensive drugs such as ACE inhibitors29 and angiotensin receptor blockers30 may enhance ACE2 expression at the cell surface and, therefore, ultimately supply SARS‑CoV‑2 with a larger number of “anchors” for infecting cells. This is still a matter of contentious debate, but it cannot be excluded that some hypertensive patients undergoing renin‑angiotensin‑aldosterone system inhibition, especially those taking ACE inhibitors, may be more susceptible to SARS‑CoV‑2 infection, which would ultimately translate into a higher risk of adverse local (ie, ARDS) or systemic (ie, SIRS and/or MOF) consequences of COVID‑19.31 On the other hand, some authors argued that hypertensive patients may experience a decreased expression of ACE2. Binding to the ACE2 receptors, SARS‑CoV‑2 attenuates residual ACE2. This leads to elevated angiotensin II levels, which drives the development of ARDS.32 Moreover, evidence convincingly attests that both pulmonary and systemic hypertension is a risk factor for unfavorable disease progression in patients with pneumonia,33 ARDS,34,35 and MOF.36 Therefore, it is plausible that the coexistence of hypertension and SARS‑CoV‑2 infection would interplay to synergistically increase the risk of unfavorable prognosis in hypertensive patients with COVID‑19 compared with those normotensive.
The recommendations for the management of hypertensive patients is another important implication of our findings. The postulated higher vulnerability to severe COVID‑19 would require specific precautions in this patient population, such as reinforcing restrictive measures for avoiding contagion (ie, strict maintenance of spatial distance of at least 1 m, social isolation, wearing more effective personal protection equipment (such as gloves, goggles, and FFP2 [N95] or FFP3 masks), as well as constant monitoring of blood pressure, to prevent particularly large changes in blood pressure, which are associated with a higher risk of developing targeted (ie, affecting the lungs) or multiple organ failure.36
A limitation of the analyzed current literature is the lack of age‑adjusted data regarding hypertension and severity of COVID‑19. In our meta‑regression analysis by the mean age of patients with severe COVID‑19, the significant OR of COVID‑19 severity associated with hypertension was only seen in patients over the age of 60 years. It is possible that the observed risk may be attributed to the higher overall disease severity and mortality in older patients, in whom the prevalence of hypertension increases with age. We hypothesize that hypertension, together with other comorbidities, influences mortality in older individuals. As such, in the coming weeks, we urgently need age‑adjusted analyses aimed to identify the clinical predictors of severe and fatal COVID‑19. Lastly, patients in the included studies who presented with no history of hypertension and in whom elevated blood pressure was noted on admission (potentially due to COVID‑19) may be considered to have a history of hypertension, which could bias the results.
In conclusion, we still lack any definitive clues to establish which comes first: the chicken (ie, hypertension) or the egg (ie, severe COVID‑19), or even to learn if these 2 conditions mutually affect each other’s pathophysiology. However, the results of this pooled analysis of the current literature would suggest that hypertension may be associated with an up to 2.5‑fold higher risk of severe and fatal COVID‑19, particularly in older individuals.
- World Health Organization. Coronavirus Disease 2019 (COVID‑19). Situation Report – 66. https://www.who.int/emergencies/diseases/novel‑coronavirus‑2019/situation‑reports/. Accessed March 27, 2020.
- Gorbalenya AE, Baker SC, Baric RS, et al. The species Severe acute respiratory syndrome‑related coronavirus: classifying 2019‑nCoV and naming it SARS‑CoV‑2. Nature Microbiology. 2020; 5: 536‑544.
- Walls AC, Park YJ, Tortorici MA, et al. Structure, function, and antigenicity of the SARS‑CoV‑2 spike glycoprotein. Cell. 2020 Mar 6. .
- Tikellis C, Thomas MC. Angiotensin‑converting enzyme 2 (ACE2) is a key modulator of the renin angiotensin system in health and disease. Int J Pept. 2012; 2012: 256294. | Crossref
- Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID‑19 infection? Lancet Respir Med. 2020 Mar 11. . | Crossref

SUPPLEMENTARY MATERIAL
ARTICLE INFORMATION